Even a short treatment with endotracheal tubes, can make the patient develop ventilator associated pneumonia (VAP), a common and very serious respiratory tract infection that can affect intubated patients.
Ventilator associated pneumonia is the second most common healthcare associated infection in intensive care, affecting up to 25% of patients.1–3 Intubation with an endotracheal tube is one important risk factor to develop a VAP, as it increases the risk that bacteria access the lower respiratory tract in many different ways.
Main causes of VAP
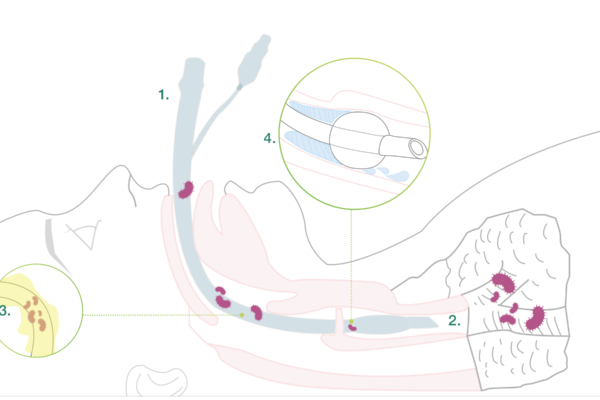
1. Intubation
During the intubation itself, there is a risk for microaspiration.
2. Impaired clearance of secretions
Intubation reduces natural defence mechanisms such as the cough reflex, which otherwise protects the lungs from secretions from the upper respiratory tract.
3. Development of a biofilm
Microbial adhesion on both the inside and the outside of the tube resulting in biofilm formation. The tube attracts bacteria and other microbes. Microbial adhesion on the tube resulting in biofilm formation is a contributing factor to infections. The bacteria might come from the patient herself or from external sources, such as personnel, other patients or medical devices. Bacteria forming biofilm are more resistant to the patient’s immune system and antibiotics.
4. Subglottic secretions
Secretions that accumulate above the cuff represent an ideal growth medium for microbes. The contaminated secretions might pass down the sides of the cuff into the lower respiratory tract.
Early/late onset VAP
Ventilator associated pneumonia is usually divided into early and late onset infection:
- Early onset VAP: Onset within four days after hospitalization. Often caused by community acquired bacteria such as pneumokocker, H. influenzae, S. aureus and antibiotic-sensitive gram-negative bacteria.
- Late-onset VAP: Onset after ≥ 5 days hospitalization. Often caused by more resistant bacteria such as P. eruginosa, Acinetobacter and Enterobacter.
Prevent VAP
It is important to prevent VAP since it leads to increased morbidity, mortality and suffering for the patients. It is also associated with a prolonged hospital stay and higher costs. On average a patient who has developed VAP stays 6.1 additional days in the ICU and 11.5 additional days in the hospital compared to a patient without VAP.4
Altogether, the attributable cost for a VAP infection is estimated to about 25 000 Euro per case.5
Guidelines to prevent VAP
- Keep the head of the patient’s bed raised
- Check the patient’s ability to breathe on his or her own
- Hand hygiene
- Clean the inside of the patient’s mouth on a regular basis
- Reduce micro aspiration (=subglottic secretion drainage)
- Limit bacterial colonization
New study shows a 53% reduction of VAP with Bactiguard’s endotracheal tube
A new Belgian study published in the renowned journal Annals of Intensive Care, shows that the risk of VAP in intensive care patients is reduced by 53% when using Bactiguard’s endotracheal tubes with subglottic secretion drainage (BIP ETT Evac).7 Read more about the study.
Read more about Bactiguard’s infection prevention endotracheal tube BIP ETT >>>
The unique Bactiguard technology reduces microbial adhesion on the endotracheal tube surface
The Bactiguard technology (Bactiguard Infection Protection) is based on a very thin noble metal alloy coating, firmly attached to the endotracheal tube surface. When in contact with fluids the noble metals create a galvanic effect, which reduces microbial adhesion. This means that less bacteria adhere to Bactiguard’s endotracheal tube, which reduces the risk of biofilm formation leading to infection.
References:
1. Ibrahim EH et al. Chest. 001;120(2):555–561.
2. Craven DE et al. Infect. 1996;11(1):32–53.
3. Rello J et al. Chest. 2002;122(6):2115–2121.
4. Rello J, Ollendorf DA, Oster G, et al. Epidemiology and outcomes of ventilator-associated pneumonia in a large US database. Chest 2002;122:2115–21.
5. Hyllienmark P. Hospital-acquired pneumonia in intensive care patients [avhandling]. Stockholm: Karolinska institutet; 2013.
6. Damas, P., Legrain, C., Lambermont, B. et al. Prevention of ventilator-associated pneumonia by noble metal coating of endotracheal tubes: a multi-center, randomized, double-blind study. Ann. Intensive Care 12, 1 (2022). https://doi.org/10.1186/s13613-021-00961-y
