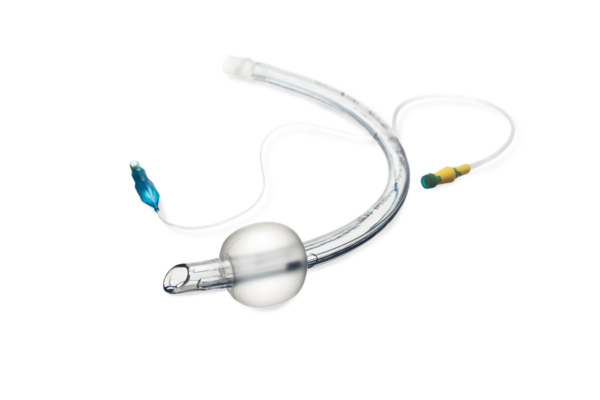
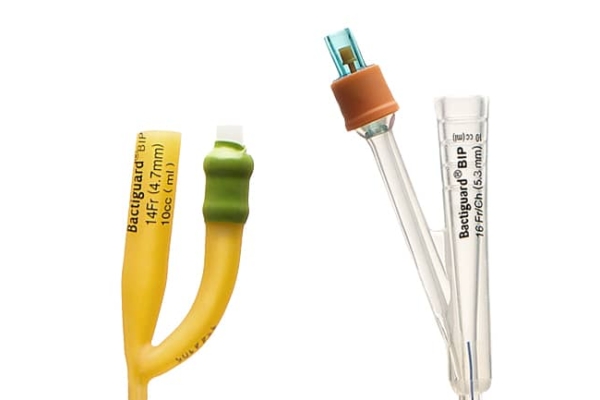
Bactiguard was founded in 2005, but the technology is almost a hundred years old.
2023
Bactiguard receives first MDR product approval, for BIP Foley Catheter.
Embarks on sharpened strategy with full focus on Licensing.
2022
Bactiguard and Zimmer Biomet expand global license partnership.
2021
Bactiguard-coated Zimmer Biomet trauma implants receive European regulatory clearance and are launched in select EMEA markets.
Bactiguard Wound Care system is launched in select markets in Europe.
Aniocyn - a new wound care range for pets is launched in the Nordic countries together with Musti Group.

2020
Acquisition of Malaysian Vigilenz Medical Devices.
Launch of BIP Foley TempSensor, a urinary catheter which combines continuous temperature monitoring with Bactiguard’s infection prevention technology.
Launch of Hydrocyn Advanced disinfectant, a non-alcoholic disinfectant for skin and surfaces, proven effective against the new Coronavirus.

2018
Orthopedic trauma implants in titanium with Bactiguard’s infection prevention technology receive the CE mark and can thus begin to be sold and used in healthcare in markets that apply EU quality standards.
License agreement is made with China’s largest manufacturer of medical devices, Well Lead for urinary catheters in China.
The BIP Foley catheter is launched in two new variants; Female and Tiemann, while the venous catheter BIP CVC is launched with Raulerson syringe.
2014
In June 2014, Bactiguard was floated on the stock exchange through an IPO, with the purpose of refinancing the company and enabling market expansion. The shares are listed and traded on the NASDAQ Stockholm under the stock symbol BACTI B.
At the end of 2014, the head office will be moved to a newly built facility on Alfred Nobels Allé in Tullinge, south of Stockholm. The new office makes it possible to gather all skills and functions such as sales and marketing, staff functions, product development and production under one roof.
The establishment in Karolinska Institutet Science Park also promotes increased collaboration and proximity to research and education at Karolinska Institutet and the KTH Royal Institute of Technology, as well as with healthcare at Karolinska University Hospital.

2006
In 2006, a development and production unit was established in Markaryd, in the south of Sweden, to focus on the further development of the technology.
The same year, work begins with developing a new product portfolio, Bactiguard Infection Protection (BIP).
1994
In 1994, the US Food and Drug Administration (FDA) approves Bard's urinary catheters Bardex IC and Lubrisil IC with Bactiguard coating (FDA 510k approval). The following year, the catheters are launched on the American market and today they are the market leaders in the USA and Japan.
1990
The company enters a partnership with the US medical device company BD (Becton, Dickinson & Company), previously C.R. Bard.
1912
The Bactiguard technology stems from the Swedish Nobel Prize laureate in in physics 1912, Gustav Dahlén, the man behind the famous AGA Lighthouse. Gustav Dahlén had an apprentice called Axel Bergström, who further developed the technique of applying a thin layer of metals to non-conductive materials. Axel then passed this knowledge on to his apprentice, Billy Södervall.