How Hydrocyn aqua works
Hydrocyn aqua mimics body’s own immune response without compromising the saftey whilst simultaneously improving wound healing.
Promotes natural wound healing
- Soothes and creates a moist wound environment to accelerate the wound healing process
Safe
- Biocompatible, non-irritant and noncytotoxic to the skin
- pH neutral and hypoallergenic
- Contains neither preservatives or parabens, nor antibiotics or steroids
- Clinically proven safe and effective
Easy to use
- Ready to use, no dilution required
- Can be combined with other forms of wound treatments
- No known contraindications
Hydrocyn aqua solution/spray
Cleans the wound, reduces bacterial load and supports the natural healing. Removes biofilm and reduces fetid odour. The active ingredient, hypochlorous acid (HOCl), is the same substance that the immune cells use to fight infections.
How to use the solution?
Clean the wound by applying the solution liberally directly to affected area. To facilitate debridement and to remove biofilm, soak dressing with the solution and leave on the wound for about 15 minutes.
Hydrocyn aqua Gel/Gel Spray
Moistens the wound and has a soothing and bacteriostatic effect. Maintaining a moist wound environment enhances the debridement process of necrotic tissue and accelerates the healing.
How to use the Gel/Gel spray?
Clean the wound with Hydrocyn aqua Solution. Apply the gel evenly to the affected area. Keep the Gel Spray 10-15 cm from the wound bed for more even application. Cover with a suitable dressing. Apply the gel in between dressing changes to maintain a moist wound environment.
Wound Healing properties
Hydrocyn aqua combines the benefits of antiseptic wound cleansing and saline/water, by eliminating pathogens without adversely affecting the tissue while maintaining a moist wound healing. In addition, hypochlorous acid, being a molecule of our innate immune system, has in several studies shown to have immunomodulatory functions that accelerate wound healing. For example, hypochlorous acid stimulates fibroblast cell-proliferation and migration1,2 and activates the transcription factors TGFß and FGF2, both known to stimulate wound healing and tissue regeneration.1
Studies also show anti-inflammatory properties2 and that hypochlorous acid contributes to increased oxygenation in the wound3.
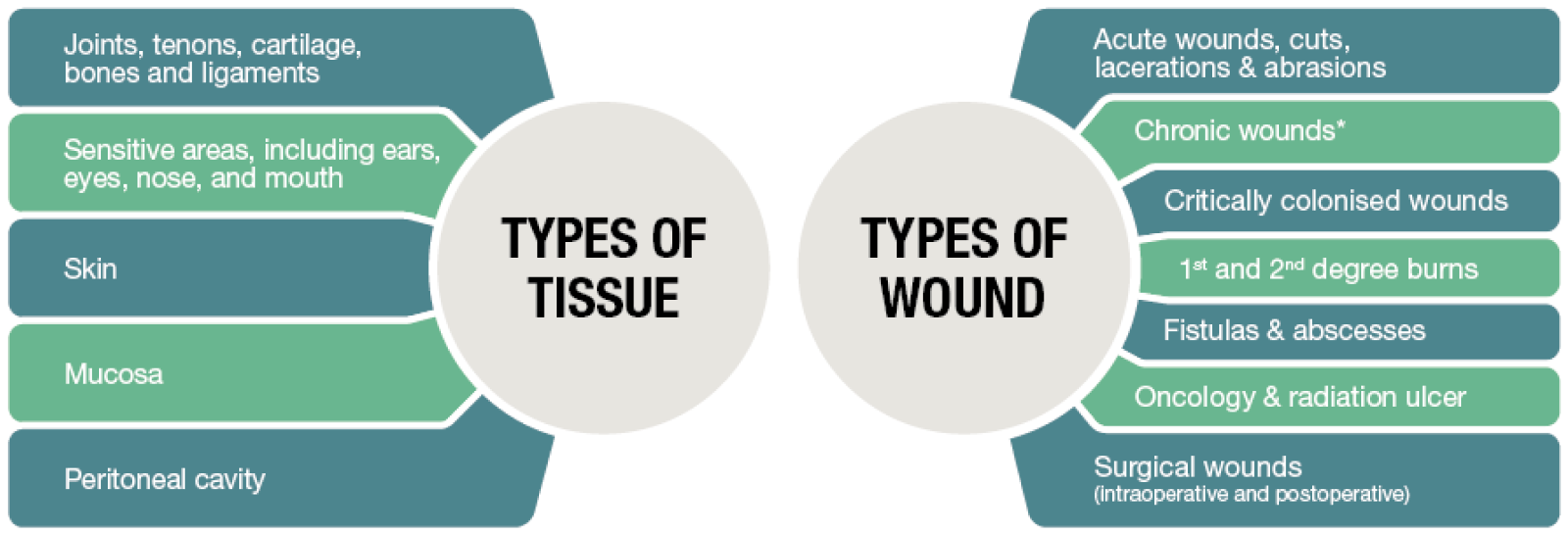
Where can Hydrocyn aqua be used?
Test results
Antimicrobial efficacy
Hydrocyn aqua Solution has been tested on several microorganisms following the ISO 11737 & ASTM E2315
standard tests. The tests show that the product is effective against both gram-positive and gram-negative bacteria
(including MRSA), fungi and spores. Test results >>>
Biocompatibility
Hydrocyn aqua Solution has been tested for biocompatibility as per ISO 10993 & has passed following in-vivo and in-vitro studies. Test results >>>
Removes biofilm
In vitro tests show that Hydrocyn aqua Solution reduces biofilm concentration up to 99.9% (Pseudomonas aeruginosa and MRSA). Test results >>>
Product range
Hydrocyn aqua system is CE marked (approved in the EU, Notified Body No 2292) medical devices class III and class IIb and meets the US Pharmacopoeia 41 NF 36, Chapter 51 – Antimicrobial Effectiveness Test.
The assortment may differ between markets, please contact us for more information. In some selected markets the products are registered under the brand Bactiguard Wound Care.
Hydrocyn aqua | |
| Product type | Packing Quantity |
| Solution Bottle Spray Mist 100ml | 50 units/box |
| Solution Bottle Spray Mist 250ml | 24 units/box |
| Solution Bottle Cap Closure 250ml | 24 units/box |
| Solution Bottle Cap Closure 500ml | 12 units/box |
| Solution Bottle Cap Closure 1000ml | 6 units/box |
| Solution Irrigation 500ml | 12 unit/box |
| Solution Irrigation 1000ml | 6 units/box |
| Gel Spray 50g | 50 units/box |
| Gel Spray 100g | 42 units/box |
| Gel tube long nozzle 15g | 20 units/box |
| Gel tube long nozzle 30g | 20 units/box |
| Gel tube long nozzle 50g | 12 units/box |
| Gel tube long nozzle 100g | 6 units/box |
| Gauze, 8-Ply 10x10cm | 25 units/box |
References:
1. Calderon-Mendoza et al. J Dent Res Vol #(Spec Iss _2015 ): Final Presentation ID #3836
2. Del Rosso et al. The Journal of clinical and aesthetic dermatology vol. 11,11 (2018): 36-39.
3. Bongiovanni CM. J Am Coll Clin Wound Spec. 2016;6(3):32-37.

